How Medicare is changing for tech
Technology is making a real difference in how we approach senior care. We’re seeing a move away from simply reacting to health crises, and toward preventative tools that help older adults maintain their independence and quality of life. This includes everything from simple medication reminders to sophisticated robots designed to prevent falls.
For a long time, Medicare coverage for these types of technologies lagged behind. The system was built around traditional medical devices – things like wheelchairs and walkers. Getting approval for newer, more proactive solutions has often been a slow and frustrating process. It’s a challenge because innovation happens quickly, and Medicare needs to adapt.
Medicare is finally updating its policies. By 2026, we expect the agency to expand coverage for more devices as they broaden the definition of what counts as 'necessary' care. It's a shift from just fixing broken bones to helping people stay independent.
The goal isn’t just to treat illness, but to help people stay healthy and avoid needing expensive interventions down the road. This shift requires a new way of thinking about what Medicare covers, and it's a process that is still unfolding. I believe it's a positive step, but it's also one that requires careful attention to detail.
The basics of durable medical equipment
At the heart of Medicare coverage for medical devices is the concept of Durable Medical Equipment, or DME. This is a broad category that includes equipment designed to withstand repeated use and is primarily for medical purposes. It's the base upon which much of Medicare's device coverage is built.
To qualify as DME, a device generally needs to meet three key criteria. First, it must be medically necessary—meaning it’s required for your health condition. Second, it must be used in your home. And third, it must be prescribed by a doctor. These aren’t just suggestions; they’re firm requirements for coverage.
Think of a traditional wheelchair, a walker, or a hospital bed. These are all examples of DME that Medicare routinely covers. They’re medically necessary, used at home, and require a physician’s order. The coverage isn't automatic, though. It's often subject to prior authorization and specific requirements.
Newer tech often struggles to fit the DME definition. A brain training app is helpful, but it isn't 'equipment' like a wheelchair. This is why the RAPID pathway exists—to handle things that don't look like traditional hardware.
- Wheelchairs for mobility
- Walkers for stability
- Hospital beds: Offering adjustable comfort and safety.
- Oxygen equipment: For respiratory support.
Getting faster approval through RAPID
The Centers for Medicare & Medicaid Services (CMS) and the Food and Drug Administration (FDA) introduced the RAPID (Revolutionizing Access to Innovative Devices) Coverage Pathway to address the slow pace of Medicare coverage for breakthrough medical technologies. This pathway is designed to accelerate access to devices that offer significant benefits to patients.
Essentially, RAPID creates a streamlined process for evaluating and covering these technologies. Devices that qualify for RAPID coverage undergo a focused review, allowing CMS to make a coverage decision more quickly. This is particularly important for devices that address unmet medical needs or offer substantial improvements over existing treatments.
To qualify for RAPID coverage, a device must meet specific criteria. It needs to be designated as a "Breakthrough Device’ by the FDA, meaning it has shown promising early evidence of effectiveness. It must also address a condition with significant unmet medical need and offer a substantial improvement over existing options. It"s a high bar to clear.
The benefits for seniors are clear: faster access to potentially life-changing technologies. However, the RAPID pathway isn’t without its critics. Some argue that it may lead to coverage of devices with insufficient evidence of long-term effectiveness. There's also debate about the criteria for determining "substantial improvement". Despite those concerns, it represents a significant step toward modernizing Medicare coverage.
What Medicare will likely cover in 2026
Looking ahead to 2026, several key technology categories are poised for expanded Medicare coverage. The changes aren’t uniform, and coverage will vary depending on the specific device and its intended use. Here's a breakdown of what we can expect.
Remote Patient Monitoring (RPM) devices are likely to see significant expansion. This includes devices like blood pressure cuffs, glucose monitors, and weight scales that transmit data to healthcare providers. Currently, Medicare covers RPM for certain conditions, like heart failure and chronic obstructive pulmonary disease (COPD). In 2026, we can anticipate broader coverage for a wider range of chronic conditions.
Fall Detection Systems are becoming increasingly important as the senior population grows. These systems use sensors to detect falls and automatically alert caregivers or emergency services. While some basic fall detection devices are currently covered as DME, more advanced systems with features like GPS tracking and two-way communication are likely to gain coverage under the RAPID pathway.
Medication Management Systems aim to improve medication adherence and reduce errors. These systems can include automated pill dispensers, medication reminder apps, and smart pill bottles. Coverage for these systems is currently limited, but we may see expanded coverage in 2026, particularly for patients with complex medication regimens.
Cognitive Support Tools, such as brain training apps and memory aids, are a more challenging area. Medicare is hesitant to cover these tools due to a lack of conclusive evidence of their effectiveness. However, if these tools demonstrate significant clinical benefits through rigorous research, they may become eligible for coverage.
Eldercare Robots are perhaps the most futuristic technology on this list. Robots like the UBTECH robot mentioned in the MIT News article are designed to assist with daily tasks, provide companionship, and even help prevent falls. While full coverage for these robots is unlikely in 2026, the RAPID pathway could open the door for limited coverage for specific models that demonstrate a clear clinical benefit. The MIT article highlights a robot that helps people sit, stand, and prevents falls—this type of device has the highest potential for coverage.
Medicare Coverage Outlook for Emerging Senior Technology (2026)
| Technology Category | Current Coverage Status | 2026 Coverage Likelihood | Key Coverage Considerations | Potential Limitations |
|---|---|---|---|---|
| Remote Patient Monitoring (RPM) | Increasingly covered for chronic conditions like heart failure and diabetes, requiring specific CPT codes. | High | Requires a physician's order, established relationship with a provider, and data transmission for qualified clinical decision-making. | Coverage generally limited to specific diagnoses and may not include all RPM devices. |
| Fall Detection Devices (Wearable/Home-Based) | Limited coverage. Some coverage exists for fall detection *integrated* with emergency response systems. | Medium | Demonstrated medical necessity, integration with a monitored emergency response system, and potential for preventing serious injury. | Standalone fall detection devices without a direct link to emergency services are unlikely to be covered. |
| Medication Management Systems (Smart Pill Dispensers) | Coverage is rare, typically requiring a documented cognitive impairment and a physician's prescription. | Low to Medium | Requires a clear link to improved medication adherence and prevention of adverse health events; documentation of cognitive decline is crucial. | Cost of devices and ongoing subscription fees may be a barrier to coverage. |
| Cognitive Training Programs (Digital Games/Apps) | Generally not covered as these are considered wellness activities, not medical treatments. | Low | Lack of robust clinical evidence demonstrating significant and lasting cognitive benefits; viewed as recreational rather than therapeutic. | Difficulty establishing medical necessity and demonstrating quantifiable improvements in cognitive function. |
| Eldercare Robotics (Assistive Robots) | Emerging area with very limited coverage. Coverage possible for specific functions like mobility assistance, but highly dependent on individual circumstances. | Low to Medium | Demonstrated functional improvement, physician's prescription, and potential to reduce the need for caregiver support. | High cost of robots, limited long-term data on effectiveness, and concerns about safety and usability. |
| Hearing Aids | Medicare Part B covers diagnostic hearing tests. Coverage for hearing aids themselves is limited to specific circumstances and is not comprehensive. | Medium | Requires a prescription and fitting by an audiologist. Coverage is primarily through Medicare Advantage plans, which vary widely. | Significant out-of-pocket costs for hearing aids even with some Medicare coverage. |
Qualitative comparison based on the article research brief. Confirm current product details in the official docs before making implementation choices.
How to handle prior authorization
Even if a technology is covered by Medicare, it often requires prior authorization. This means your doctor must obtain approval from Medicare before you receive the device. It’s a crucial step, and failing to get prior authorization can result in denied claims and out-of-pocket expenses.
Prior authorization is required to ensure that the device is medically necessary and meets Medicare’s coverage criteria. Your doctor will need to submit documentation, including a prescription, medical records, and a detailed justification of why the device is necessary for your specific condition. The more thorough the documentation, the better.
Common reasons for denial include insufficient documentation, lack of medical necessity, or the device not meeting Medicare’s coverage requirements. If your request is denied, you have the right to appeal. The appeal process can be complex, so it’s important to understand your rights and gather all relevant information.
To increase your chances of approval, work closely with your doctor to ensure the documentation is complete and accurate. Be prepared to provide additional information if requested by Medicare. Don’t hesitate to contact your SHIP center (State Health Insurance Assistance Program) for assistance. They can provide guidance and support throughout the prior authorization process. It’s a bureaucratic hurdle, but it’s often worth the effort.
Resources and Support
Navigating Medicare coverage for senior technology can be complex. Fortunately, there are many resources available to help you understand your options and get the support you need.
The official Medicare website () is a great place to start. You can find information about coverage, eligibility, and the prior authorization process. The CMS and FDA RAPID coverage pathway information can be found at
SHIP (State Health Insurance Assistance Program) centers offer free, unbiased counseling to Medicare beneficiaries. You can find your local SHIP center by visiting Several advocacy organizations, such as the National Council on Aging (), provide information and support for seniors and their families.
Essential Technology for Seniors: Medicare-Eligible Devices in 2026

Clinically validated for accuracy · Recommended by doctors and pharmacists · Designed for home use with an upper arm cuff
This blood pressure monitor is a common and recommended device for home health monitoring, a category often considered for Medicare coverage.
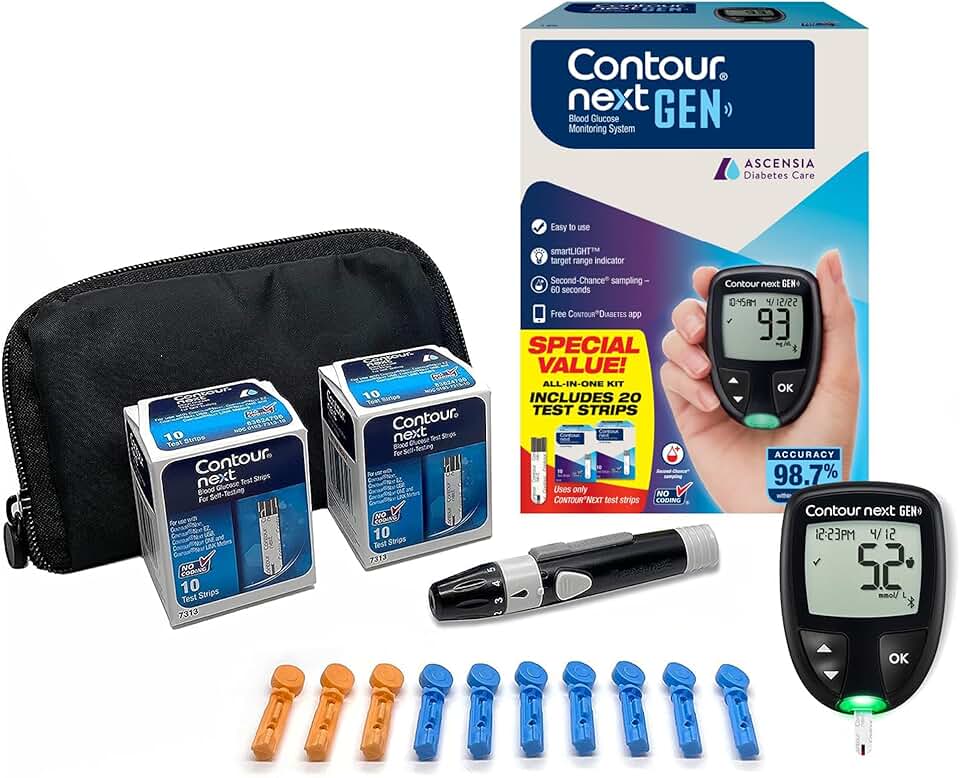
All-in-one kit with 20 test strips · Accurate blood sugar monitoring · Portable and easy-to-use design
Blood glucose monitors are essential for diabetes management and represent a category of medical devices that may be eligible for Medicare coverage.

Automatic fall detection · No monthly or activation fees · AI technology for accuracy
Personal safety devices with fall detection are increasingly important for seniors, and this type of technology is a potential area for Medicare coverage.

28-day capacity with pill dispensing · Upgraded LCD display and key lock · Sound and light alerts for medication reminders
Automatic pill dispensers help ensure medication adherence, a critical aspect of senior health management that may be considered for Medicare coverage.
As an Amazon Associate I earn from qualifying purchases. Prices may vary.





No comments yet. Be the first to share your thoughts!